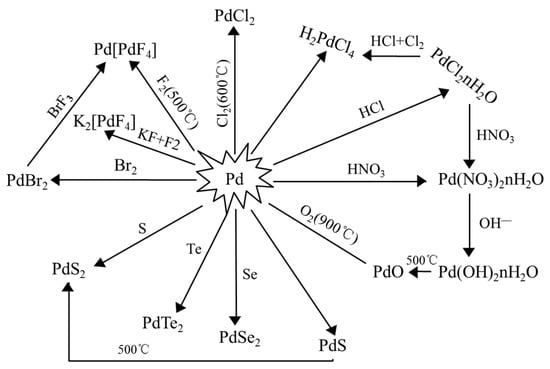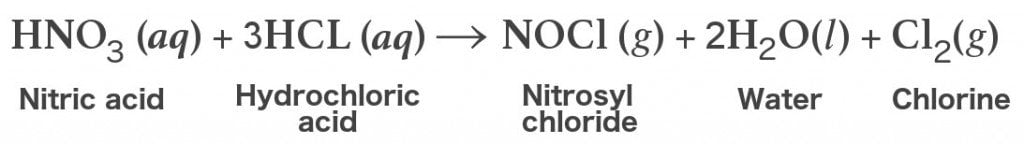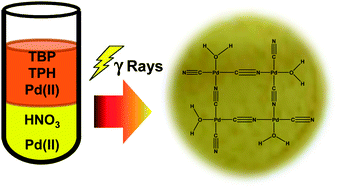
Characterization of palladium species after γ-irradiation of a TBP–alkane–Pd(NO3)2 system - RSC Advances (RSC Publishing)

of residue obtained after leaching nitric acid residue with aqua-regia... | Download Scientific Diagram

Recovery of palladium(II) from strong nitric acid solutions relevant to high-level liquid waste of PUREX process by solvent extraction with pyrazole-pyridine-based amide ligands - ScienceDirect
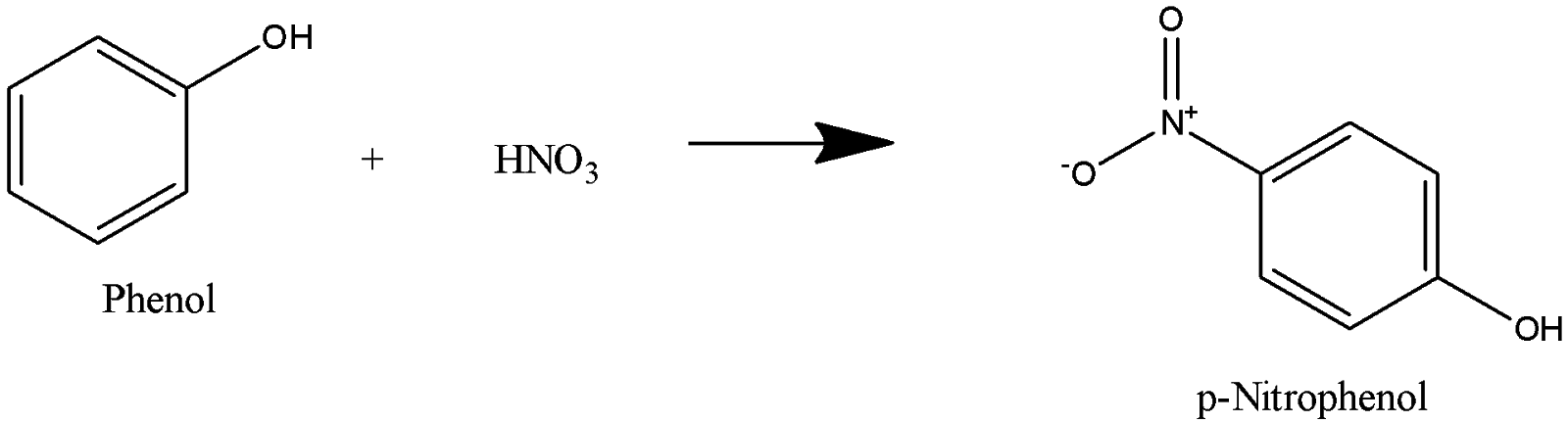
Which of the following sets of reactants is used for the preparation of paracetamol from phenol?(A) $HN{{O}_{3}},{{H}_{2}}\/Pd,{{(C{{H}_{3}}CO)}_{2}}O$(B) ${{H}_{2}}S{{O}_{4}},{{H}_{2}},Pd,{{(C{{H}_{3}}CO)}_{2}}O$(C) ${{C}_{6}}{{H}_{5}}{{N}_{2}}Cl,SnC ...
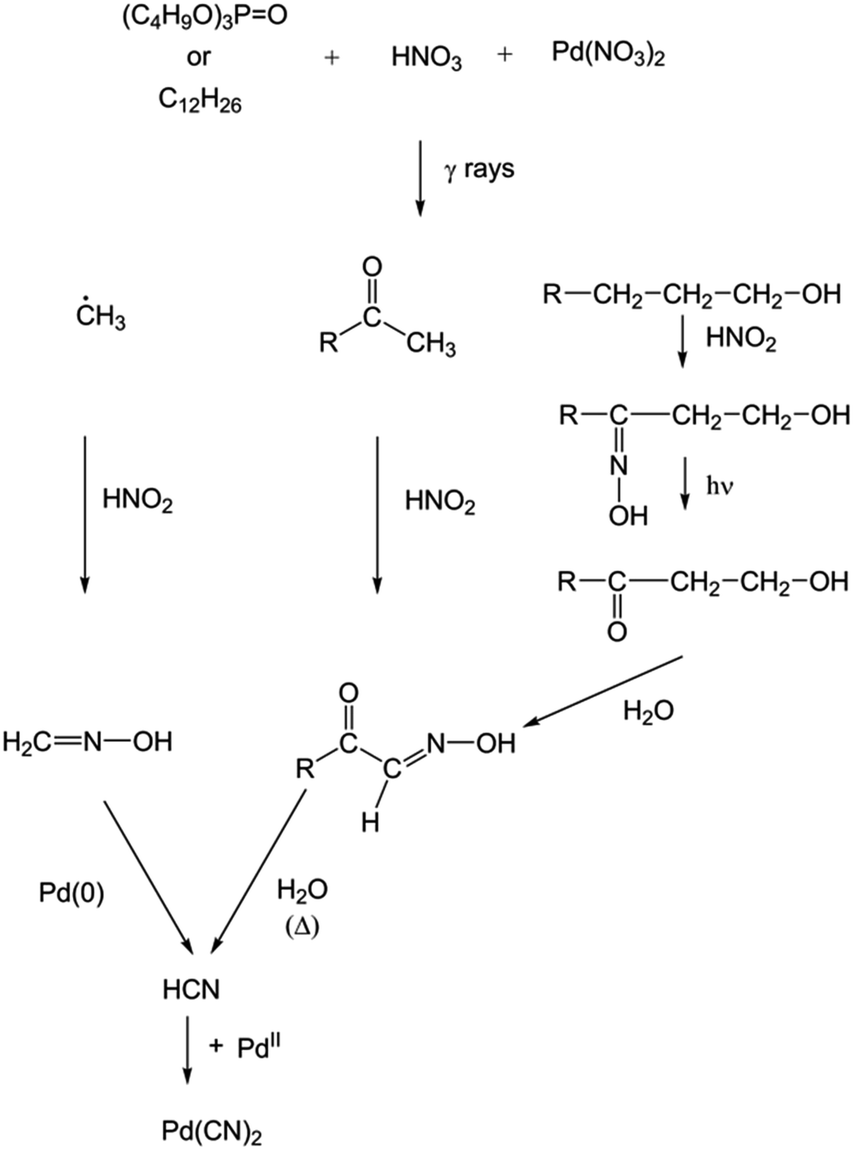
Characterization of palladium species after γ-irradiation of a TBP–alkane–Pd(NO 3 ) 2 system - RSC Advances (RSC Publishing) DOI:10.1039/C8RA02695E
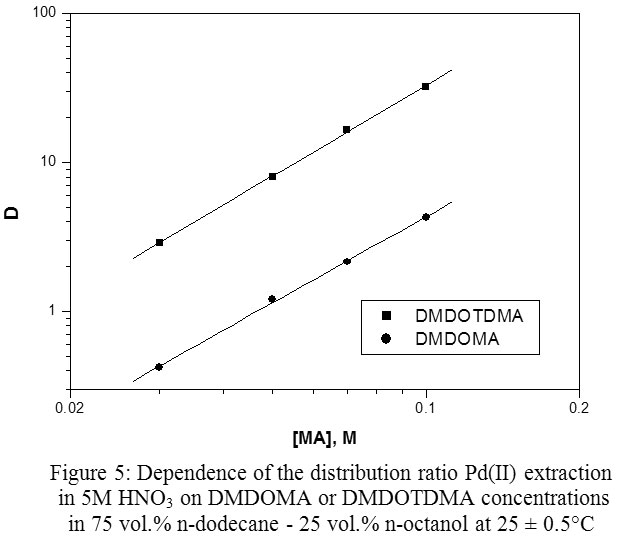
Recovery of Palladium from Concentrated Nitrate Solutions with N,N′‐Dimethyl‐N,N′‐Dioctyltetradecylmalonamide as new Extractant : Oriental Journal of Chemistry
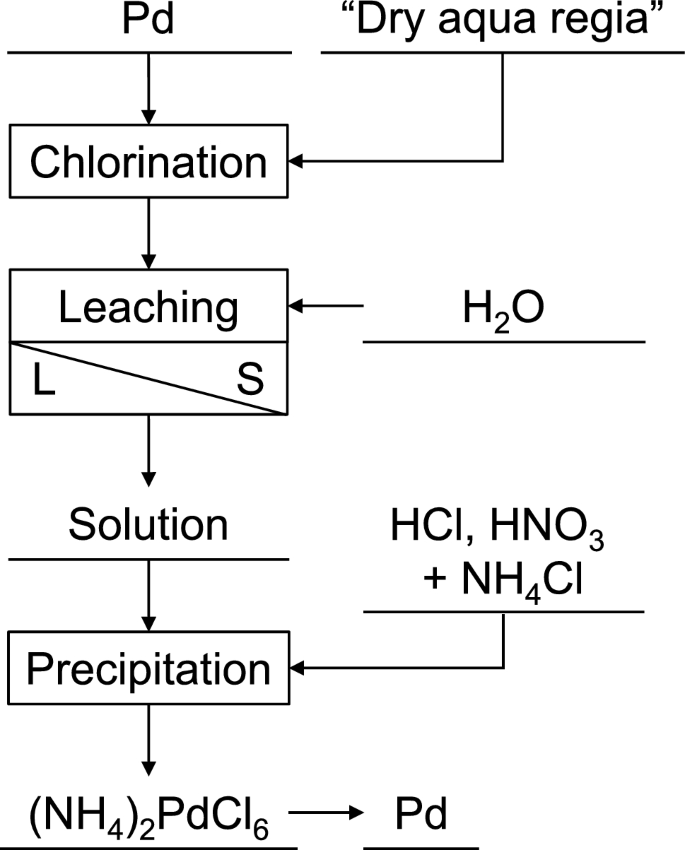
Fundamental Study of Palladium Recycling Using “Dry Aqua Regia” Considering the Recovery from Spent Auto-catalyst | SpringerLink

Recovery of Palladium(II) from nitric acid medium using a natural resin prepared from persimmon dropped fruits residues - ScienceDirect

Palladium(II) nitrate solution 10 wt. % in 10 wt. % nitric acid, 99.999% trace metals basis | Sigma-Aldrich
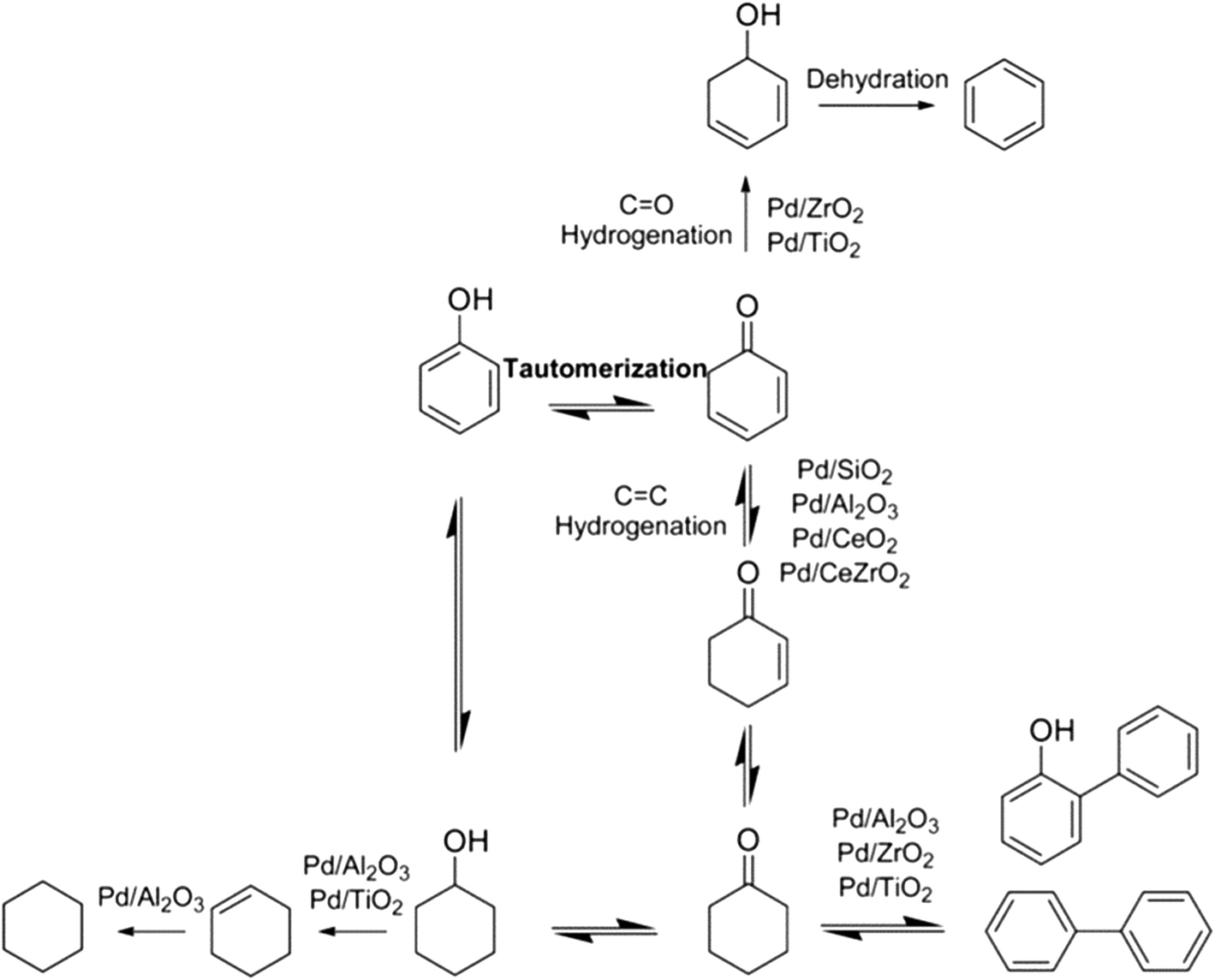
Towards high-performance heterogeneous palladium nanoparticle catalysts for sustainable liquid-phase reactions - Reaction Chemistry & Engineering (RSC Publishing) DOI:10.1039/D0RE00197J

Electrochemical Synthesis of Nitric Acid from Nitrogen Oxidation - Wang - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Palladium(II) nitrate solution 10 wt.% in 10 wt. % nitric acid, 99.999% Palladium(II) nitrate solution 10 wt.% in 10 wt. % nitric acid, 99.999% Manufacturers, Suppliers, Price | India, China